A Canadian company is pushing forward to developed to indicate a positive result only when the Covid-19 virus is present, allowing for direct and clear interpretation. When completed, this test Covid-19 test is expected to produce results in 5-15 minutes and is anticipated to cost less than $50. It will not require any additional laboratory-based equipment and can be administered by a layperson at the point of care.
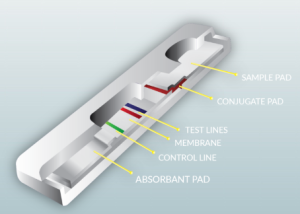
Until now the majority of testing completed for Covid-19 uses molecular-based technology (PCR), a testing platform that typically costs more than $200 per test, frequently takes 2-4 hours to produce results, and requires specialized laboratory equipment and skilled technicians to operate.
To date, the only competitive lateral flow tests that have been announced for sale are serological assay tests, which are designed to identify IgM and IgG antibodies present post infection.
Serological tests are susceptible to producing false positive and false negative results if a patient is suffering from any one of a variety of unrelated infections (I.e. ear or tooth infection, regular flu, etc.).
This test can be used as a screening tool to rapidly identify patients as being ‘at risk’ or ‘not at risk’ of Covid-19. This will help ease the burden on health practitioners and allow resources to be diverted to those who need it most.
use in a variety of scenarios, such as: An in-home test and monitoring To identify if patients require further testing or treatment in a clinical setting or to verify if patients are ready for release from quarantine, or to screen individuals prior to entering closed public venues such as cruise ships and airplanes.
